Introduction
Cytogenetic alterations are essential prognostic biomarkers in patients (pts) with acute lymphoblastic leukemia/lymphoma (ALL), but are limited by the propensity of leukemic blasts to undergo apoptosis in culture, poor-quality chromosomes, and overgrowth by normal cells. Array CGH + SNP has 10,000-fold higher resolution than conventional cytogenetics and can identify submicroscopic gains, losses and copy number neutral loss of heterozygosity (cnLOH) irrespective of mitotic rate. Few studies outside of clinical trials have investigated microarray findings in adults with ALL or correlated microarray findings with clinical outcomes.
Methods
We retrospectively reviewed pts with ALL and mixed phenotype acute leukemia (MPAL) aged ≥18 years at diagnosis at our institution between 2017 and 2022. Array analysis was performed on diagnostic bone marrow or peripheral blood samples using high resolution CGH + SNP Genetisure cancer array platform (Agilent). Statistical analysis was done using R software (R core team, 2022).
Results
Baseline characteristics of included pts (n = 60) are shown in Table 1. CGH + SNP analysis identified 93.3% (56/60) of pts with genomic changes with a mean of 7 (range 0 - 28) aberrations per pt. Among pts with a normal karyotype, 81.8% (9/11) had new genomic abnormalities revealed by CGH + SNP, but significantly fewer aberrations were detected per pt compared to those with abnormal karyotypes (mean 3.09 v. 7.67, p = .008). Pts with Ph-like B-ALL/MPAL had significantly more aberrations on array than pts without Ph-like phenotype (mean 12.0 v. 5.85, p.= .002).
Among the 78.3% of pts without complex cytogenetics on chromosome analysis, 44.7% (21/47) were found to have a complex genomic profile (≥5 aberrations) with the addition of CGH + SNP. However, there was no difference in CR rates or OS in pts with <5 or ≥5 microarray aberrations (p = .515 and p = .970, respectively).
Quantity of copy number alterations (CNAs) as detected on array was not predictive of achieving end of induction CR/CRi/CRh (mean 6.08 in pts who achieved CR/CRi/CRh v. 7.02 in pts with primary refractory disease, p = .310). Hispanic pts had the same number of CNAs as non-Hispanic pts (mean 6.69 v. 6.94, p = .435). On linear regression analysis, there was no correlation between number of CNAs and age (r 2 = -.01, F = 0.1941, p = .661) or BMI (r 2 = 5.21 x10 -5, F = 0.0029, p = .957).
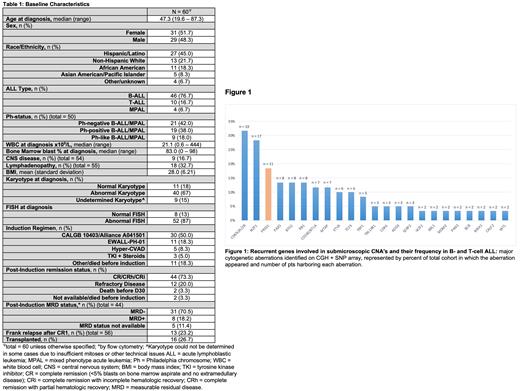
The most frequent focal aberrations were detected in CDKN2A/B (n = 19), IKZF1 (n = 17), PRSS1 (n = 11), PAX5 (n = 8), BTG1 (n = 8) and RB1 (n = 8) (Figure 1). RB1 loss was enriched in pts with T-ALL (45.5% v. 6%, p = .003),and occurred at significantly higher rates than previously reported in T-ALL (Schraders, 2009). Loss of PRSS1 was detected in 18.3% (11/60) of the cohort. Homozygous PRSS1 deletions were identified in 3/11 (27.3%) pts. We identified the minimal overlapping deleted region on 7q34 in all 11 pts, which included the PRSS1 gene within the 124 kb region. Five of 6 pts who had repeat arrays performed at the end of induction had resolution of this aberration. The one pt who retained PRSS1 loss after induction had primary refractory disease, but after further treatment and attainment of CR1, this pt cleared the PRSS1 loss. Asparaginase associated pancreatitis, which has been linked to PRSS1 variations, occurred in only 1 pt with PRSS1 loss. Pts harboring PRSS1 loss were significantly less likely to achieve MRD negativity after induction (50% vs. 82%, P = .041) and had numerically more relapses (37.5% vs. 19%, p = .300), but no OS difference was observed (p = .290).
Conclusions
In our institutional cohort, the addition of CGH + SNP array reclassified almost half (44.7%) of pts without complex cytogenetics to harbor complex genomic changes (≥5 aberrations). Pts with Ph-like ALL had significantly more aberrations on array than those with Ph-positive or -negative B-ALL. Focal PRSS1 loss was associated with reduced rates of MRD clearance at the end of induction. This aberration resolved upon disease remission, raising the possibility that both homozygous and/or heterozygous deletions of 124 kb from the 7q34 chromosomal region of the PRSS1 gene may potentially have a role in ALL pathogenesis. Further studies with larger cohorts utilizing CGH + SNP analyses at additional time points during treatment are needed to explore the potential integration of microarray data for refined ALL risk stratification as well as to elucidate the functional role of PRSS1 in leukemogenesis.
Disclosures
Feld:Oryzon: Research Funding; Gilead: Consultancy; Syros: Research Funding; Taiho: Research Funding. Tremblay:AbbVie: Consultancy; Sierra Oncology: Consultancy; GSK: Consultancy; Cogent Biosciences: Consultancy; Astellas Pharma: Research Funding; Gilead: Research Funding; Novartis: Consultancy; CTI Biopharma: Consultancy, Research Funding. Kremyanskaya:Protagonist Therapeutics, Inc.: Consultancy, Research Funding. Mascarenhas:Bristol Myers Squibb, Celgene, CTI BioPharma, Geron, Incyte Corporation, Janssen, Kartos Therapeutics, Merck, Novartis, PharmaEssentia, Roche; Participated in consulting or advisory committees - AbbVie, Bristol Myers Squibb, Celgene, Constellation Pharmac: Research Funding; Incyte, Novartis, Roche, Geron, GSK, Celgene/BMS, Kartos, AbbVie, Karyopharm, PharmaEssentia, Galecto, Imago, Sierra Oncology, Pfizer, MorphoSys, CTI Bio: Consultancy; Bristol Myers Squibb, Celgene, Constellation Pharmaceuticals/MorphoSys, CTI BioPharma, Galecto, Geron, GSK, Incyte Corporation, Karyopharm Therapeutics, Novartis, PharmaEssentia, Prelude Therapeutics, Pfizer, Merck, Roche, AbbVie, Kartos: Consultancy, Membership on an entity's Board of Directors or advisory committees; GSK: Honoraria; AbbVie, Bristol Myers Squibb, Celgene, CTI BioPharma, Geron, Incyte Corporation, Novartis, Janssen, Kartos Therapeutics, Merck, PharmaEssentia, Roche: Research Funding; AbbVie, CTI BioPharma Corp, a Sobi company, Geron, GlaxoSmithKline, Imago, Incyte, Kartos, Kayropharm, MorphoSys, Novartis, Pfizer, PharmaEssentia, Sierra: Consultancy. Bar-Natan:Incyte: Research Funding; Amgen: Research Funding; BMS: Research Funding.